Water for Injection (WFI)
Microbiology, Endotoxin and Chemical Tests
What is Water for Injection (WFI) Analysis?
Purified Water is the number one raw material used in Pharmaceutical manufacturing. Water for Injection (WFI) is a further purification of pharmaceutical purified water via distillation, this distillation step removes bacterial endotoxins from the water. WFI is primarily used in products or processes that come into direct contact with the bloodstream. It is therefore essential that the endotoxin levels are controlled and monitored as they can elicit a pyrogenic response when introduced to the bloodstream.
Why do we test it?
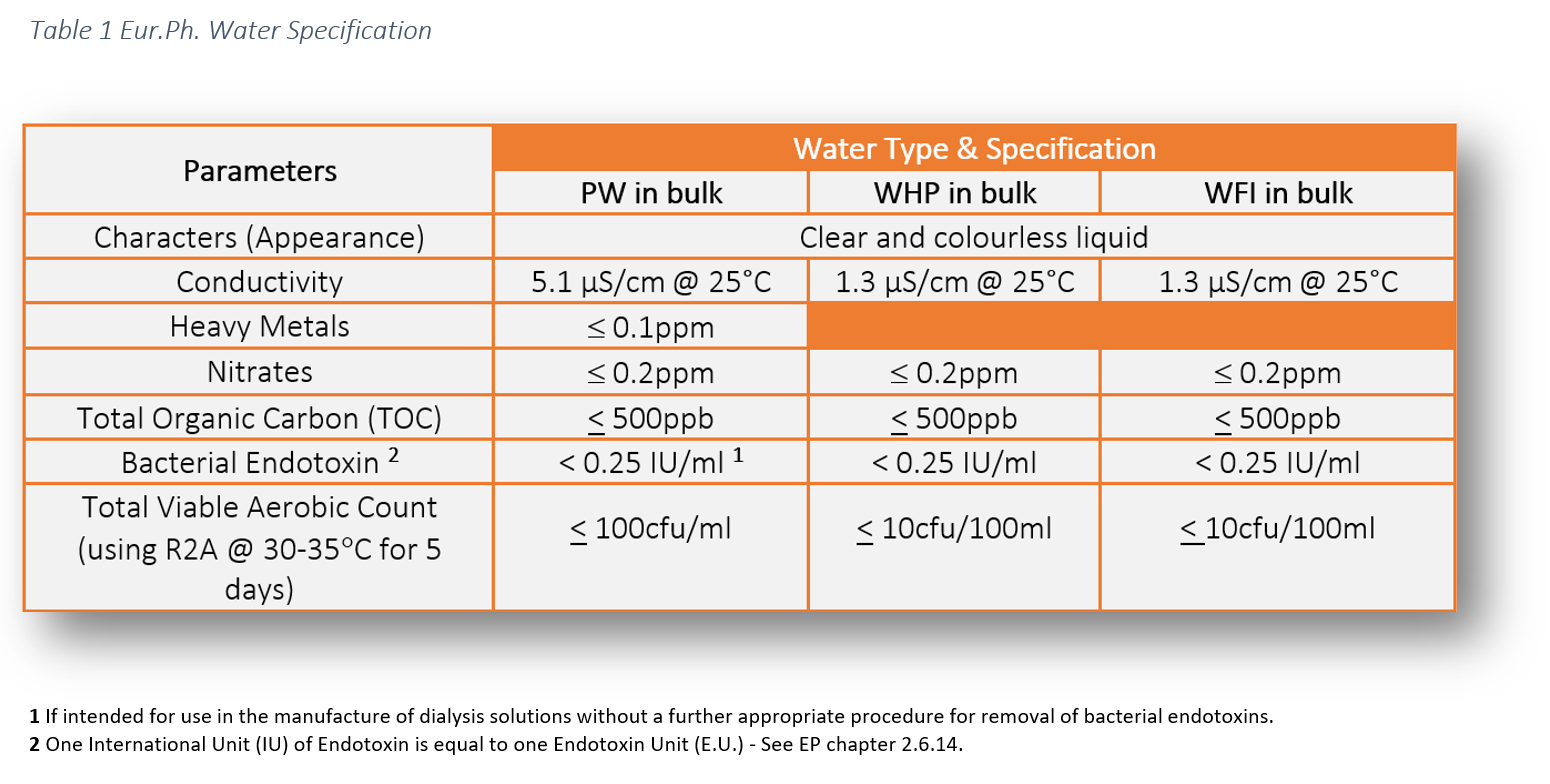
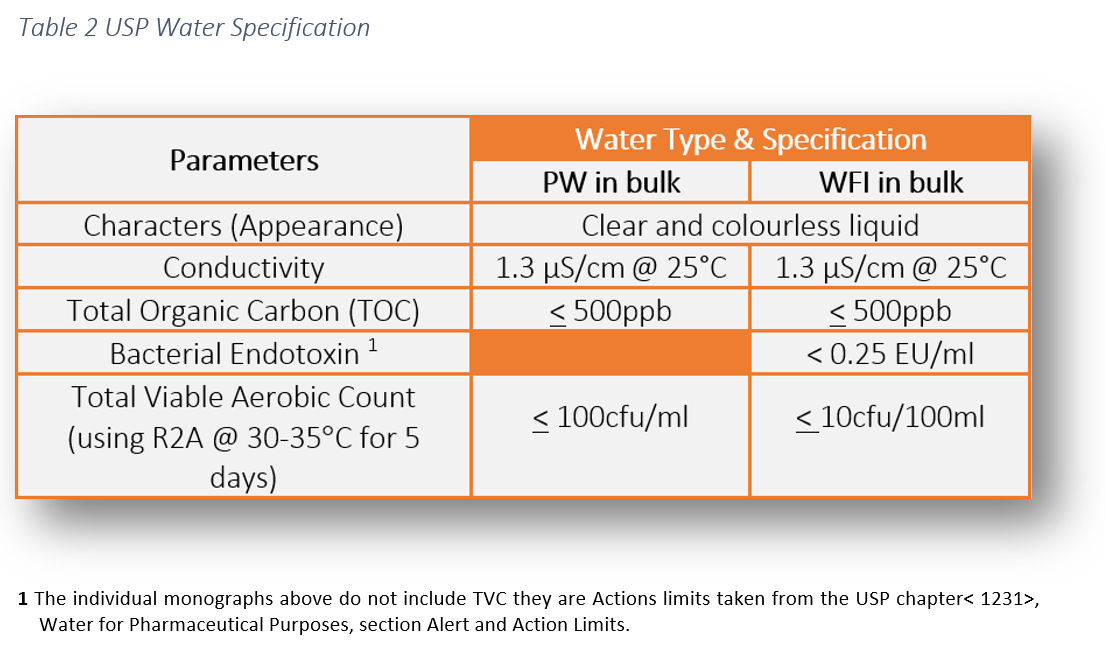
The legal requirement to comply with pharmaceutical water specifications is defined in the pharmacopoeial monographs. These water specifications can be found in the following pharmacopoeias: USP, Ph Eur and JP. This literature establishes the minimum quality standards within the industry which are enforceable by the regulators. As manufacturers and services providers we have a responsibility to ensure the highest level of operational control to ensure product safety for patients. The additional risk and extra testing associated with WFI is attributable the route of product administration.
How can we help assure compliance?
At Honeyman we are able to provide the full suite of microbiological and chemical testing to ensure compliance with the following:
- European Pharmacopoeia (Ph. Eur.)
- United States Pharmacopoeia (USP)
- Harmonized tests covering both Ph. Eur and USP (EP/USP)
- Japanese Pharmacopoeia (JP)
- In house specifications
The chemical water quality of WFI is comparable with Purified Water, however, the difference lies in the endotoxin specification.
To satisfy the microbiological and endotoxin quality component we are able to perform:
- Total viable count (TVC) via membrane filtration (10cfu/100ml limit)
- Limulus Amebocyte Lysate (LAL) turbidometric kinetic assay for the detection of endotoxins, also known as an Endotoxin test (0.25EU/ml limit)
Our suite of chemical tests to ensure compliance include:
- Conductivity
- pH
- Nitrates
- Total Organic Carbon
- Heavy metals
Water for Injection (WfI)
To Request a Quotation or Book a Test - Click Below to Contact Us:
Our Customers:
























